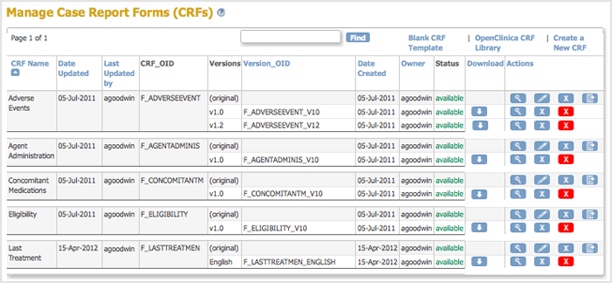
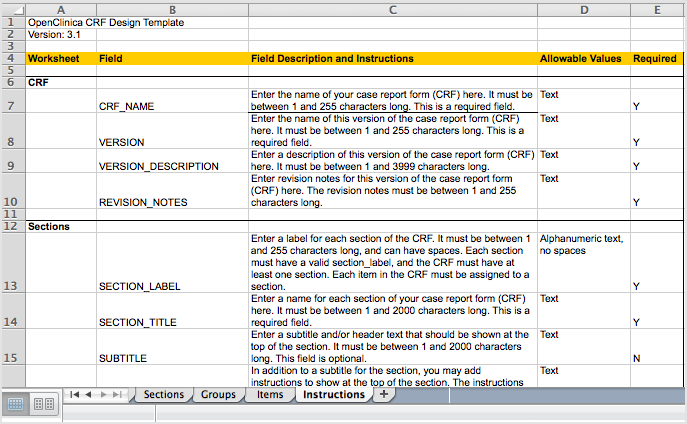
Clinical trial case identification and classification results. ABC =... | Download Scientific Diagram
Defining clinical trial quality from the perspective of resource-limited settings: A qualitative study based on interviews with investigators, sponsors, and monitors conducting clinical trials in sub-Saharan Africa | PLOS Neglected Tropical Diseases

Introduction. The Evolution of the Data Management Role: The Clinical Data Liaison - PDF Free Download

Data Management: Source Documents, Case Report Forms and Electronic Data Thomas Salerno, RN BSN MA Senior Team Lead Cardiology Research Jefferson Clinical. - ppt download

Need for aligning the definition and reporting of cytokine release syndrome (CRS) in immuno-oncology clinical trials - Cytotherapy

Case Study: Converting Paper-based Case Report Forms to an Electronic Format (e-CRF) with ACASI Self-Report Integration. - Abstract - Europe PMC